Programs
Denifanstat – Our Lead Product Candidate
Denifanstat, our lead product candidate, is an oral, once daily pill and selective fatty acid synthase (FASN) inhibitor. FASN is the key enzyme in the de novo lipogenesis (DNL) pathway that converts metabolites of dietary sugars such as fructose into palmitate, a saturated fatty acid.
Denifanstat was selected from our extensive compound library after a rigorous medicinal chemistry and preclinical development effort. Denifanstat has been studied in more than 1,200 subjects, including healthy volunteers, patients with solid tumors, patients with acne and patients with MASH.
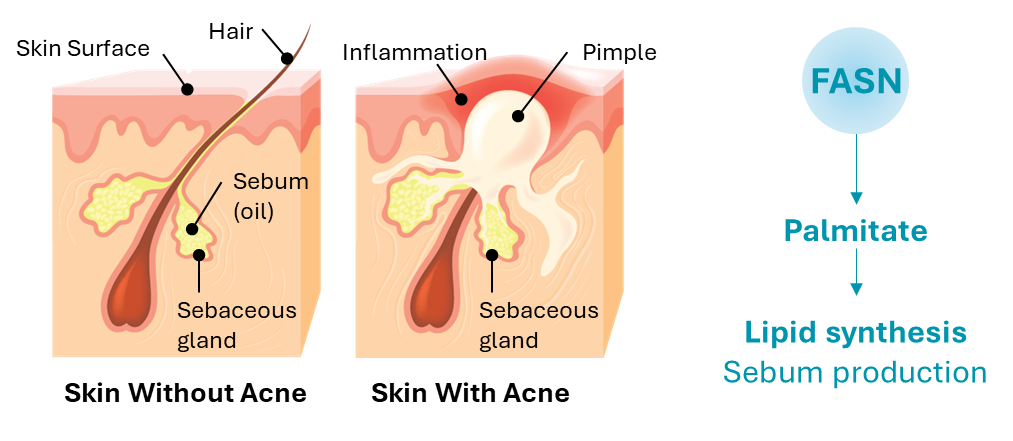
Treating Acne with FASN Inhibition
Acne is one of the most common skin conditions in the U.S., with approximately 50 million Americans affected annually and more than 5 million seeking medical treatment for acne each year. Acne affects around 85% of persons between the ages of 12 and 24. Moderate to severe acne accounts for 20% of acne sufferers, or approximately 10 million people in the U.S. annually. There is no cure for acne; and due to its pathology, most patients require chronic management and multiple annual courses of treatment for flare control.
Acne is a promising therapeutic area for application of FASN inhibitors because FASN is required for sebum production, which is upregulated in acne and leads to exacerbation of acne lesions including development of nodules and cysts.
Recent Phase 3 Clinical Trial Data Demonstrates Denifanstat’s Potential in Acne
In June 2025, we reported that denifanstat met all primary and secondary endpoints in a Phase 3 clinical trial for the treatment of moderate to severe acne vulgaris conducted by Sagimet’s license partner Ascletis Bioscience Co. Ltd. (Ascletis) in China. Ascletis reported that denifanstat was generally well-tolerated. The Phase 3 clinical trial (NCT06192264) was a randomized, double-blind, placebo-controlled, multicenter clinical trial in China to evaluate the safety and efficacy of denifanstat for the treatment of patients with moderate to severe acne. The 480 enrolled patients were randomized 1:1 into two treatment arms to receive denifanstat 50mg or placebo, once daily for 12 weeks.
In December 2025, Ascletis announced that the China National Medical Products Administration (NMPA) accepted its New Drug Application (NDA) for denifanstat for the treatment of moderate to severe acne.
In January 2026, Ascletis reported positive topline results in the open-label Phase 3 trial evaluating the long-term safety of ASC40 (denifanstat) tablets in patients with moderate to severe acne in China.
Development of Denifanstat for an Acne Indication in the US
Sagimet plans to file an Investigational New Drug (IND) Application for denifanstat for the treatment of moderate to severe acne in mid-2026. Following IND clearance, Sagimet anticipates advancing denifanstat into a registrational Phase 3 trial in moderate to severe acne patients in 2H 2026.
FASN Inhibitor TVB-3567 in Development for an Acne Indication
Our second FASN inhibitor, TVB-3567, is a potent and selective small molecule FASN inhibitor, planned to enter clinical development for the treatment of acne. TVB-3567 showed potent FASN inhibitory activity based on inhibition of palmitate synthesis in human, rat, mouse, and dog cell lines; a single dose of TVB-3567 inhibited palmitate synthesis in a rat model. These studies include the standard suite of IND-enabling, GLP-compliant safety pharmacology and genotoxicity studies, and GLP-compliant general toxicology studies of up to four weeks treatment duration in rats and dogs.
In June 2025, we initiated a first-in-human Phase 1 clinical trial of TVB-3567, for development of an acne indication. The Phase 1 clinical trial is a randomized double-blind placebo-controlled trial designed to evaluate the safety, tolerability, pharmacokinetics and pharmacodynamics of TVB-3567 in healthy participants with or without acne. The trial is comprised of several parts, including single ascending dose cohorts and multiple ascending dose cohorts in participants without acne, followed by testing in participants with acne including evaluation of pharmacodynamic biomarkers.
Upon completion of the Phase 1 clinical trial of TVB-3567, subject to consultation with regulatory authorities, Sagimet plans to initiate a Phase 2 clinical trial with TVB-3567 in moderate to severe acne patients in 2H 2026.
Clinical Data Demonstrates Denifanstat’s Potential to Treat Metabolic Dysfunction-Associated Steatohepatitis (MASH)
In FASCINATE-2, a Phase 2b clinical trial of denifanstat in biopsy-confirmed metabolic dysfunction-associated steatohepatitis (MASH) patients with moderate to advanced fibrosis (F2/F3) at week 52, denifanstat met all primary endpoints. In this trial, denifanstat, showed statistically significant improvements relative to placebo on both of the primary endpoints of MASH resolution without worsening of fibrosis with ≥2-point reduction in NAS, and ≥2-point reduction in NAS without worsening of fibrosis. Denifanstat-treated patients also showed statistically significant fibrosis improvement by ≥ 1 stage with no worsening of MASH, and a greater proportion of MRI-derived proton density fat fraction (MRI-PDFF) ≥30% responders relative to placebo.
Combination of denifanstat and resmetirom for the treatment of MASH
Pre-clinical data presented at EASL in 2024 for two mouse models of MASH showed that the combination of a FASN inhibitor (TVB-3664, a surrogate for denifanstat) and the thyroid hormone receptor beta (THR-β ) agonist, resmetirom, had a synergistic effect on important liver disease markers, including improvement of NAS by histologic analysis and more robust improvement in hepatic collagen content compared to the single agents. Synergistic activity of the combination was demonstrated in the rate of histological improvement (NAS ≥2 points), which was 33% for the FASN inhibitor monotherapy, 25% for resmetirom monotherapy, and 80% for the combination of the two, a level of improvement that greatly exceeded a simple addition of the activity of the two drugs.
We completed a Phase 1 clinical trial to evaluate the pharmacokinetics (PK) and tolerability of a combination of denifanstat and resmetirom in December 2025.
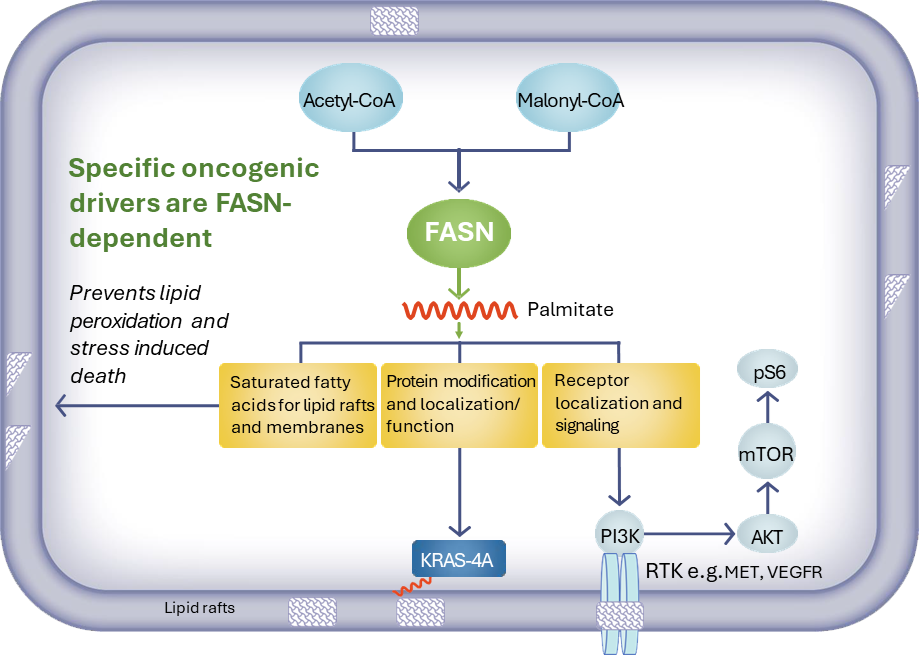
Oncology
Dysregulation of lipid metabolism is a hallmark of certain cancers. Increased expression of FASN has been associated with poor prognosis and reduced survival in tumor cell types. Several cancer types have been shown to upregulate FASN to rewire lipid metabolism and change the nature of the tumor cell membrane making these cells resistant to traditional cancer drugs. FASN inhibition can also potentially address the enormous challenge of resistance to cancer therapies.
We completed a Phase 1 clinical trial with denifanstat in 136 patients with advanced, heavily pretreated and mostly metastatic solid tumors, which demonstrated clinical activity in defined patient populations and provides the foundation for future clinical development.
We are evaluating denifanstat either alone or in combination with other classes of oncology drugs in specific subsets of solid tumors that are FASN-dependent including:
- Metastatic castration resistant prostate cancer, FASN-dependent –– Investigator-sponsored Phase 1 clinical trial of denifanstat in combination with enzalutamide therapy ongoing.
- Hepatocellular carcinoma FASN-dependent – Positive preclinical combination with kinase inhibitors, supported by translational bioinformatics.
- Non-small cell lung cancer/KRAS mutation – Positive preclinical combination results with KRAS inhibitor, encouraging results observed in patients with NSCLC KRASM tumors enrolled in a Phase 1 clinical trial in patients with solid tumors.