Pipeline
Development Pipeline: Indications and Clinical Milestones
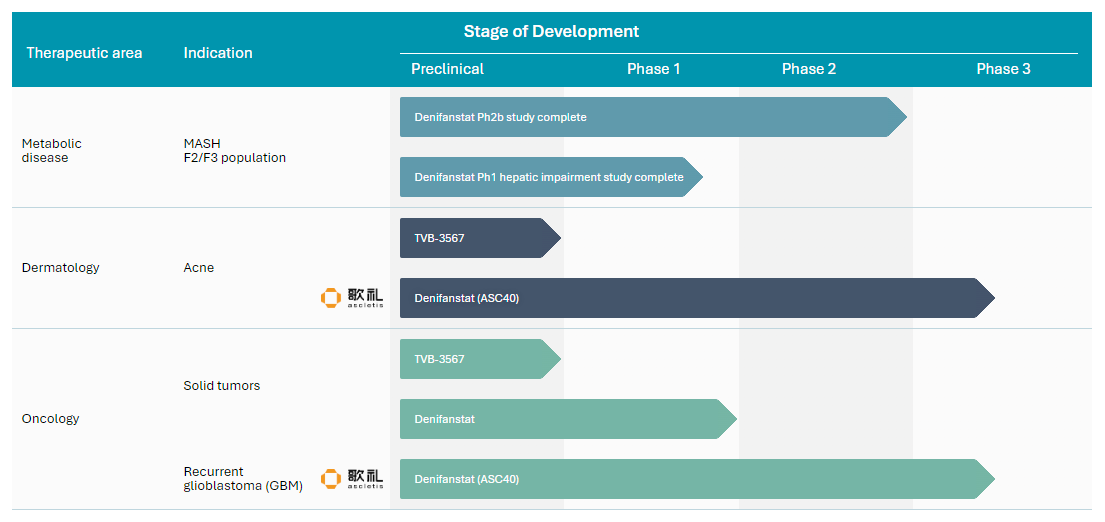
Stage of Development
Therapeutic area
Indication
Preclinical
Phase 1
Phase 2
Phase 3
Dermatology
Denifanstat Phase 3 for the US expected to initiate in 2H 2026
TVB-3567
FASNi Topical*

Denifanstat (ASC40)
Metabolic
disease
Denifanstat Phase 3 ready (F2/F3MASH)
Denifanstat Ph1 hepatic impairment study complete
Denifanstat/resmetirom Ph 1 PK trial complete
*FASN inhibitor Topical Formulation
Development Pipeline: Indications and Clinical Milestones
Stage of Development
Therapeutic area
Indication
Expected Milestone / Status
Preclinical
Phase 1
Phase 2
Phase 3
Metabolic
disease
Denifanstat
Dermatology
Denifanstat (ASC40)
TVB-3567
* Trials conducted in China by Ascletis, who has licensed development and commercialization rights to all indications in Greater China

